Artificial Photosynthesis
DEFINITION & TYPES OF STRATEGIES
Artificial photosynthesis is a field research that tries to replicate the natural process of photosynthesis with the aim of turnning carbon dioxide and water into carbohydrates and oxygen using sunlight. Current researches are focused on hydrogen catalysts, water-oxidizing catalysts, photosensitizers, carbon dioxide reduction catalysts and other materials and components.
The advantages of artificial photosynthesis fuel include:
- The solar energy can be immediately converted and stored. In photovoltaic cells, sunlight is converted into electricity and then converted again into chemical energy for storage.
- The products of the reactions are environmentally friendly. Artificially photosynthesized fuel would be a carbon-neutral source of energy, which could be used for transportation or homes.
The efficiency achieved in catalyst is the key: the average of the incident light that can be used in a system must be analyzed.
Photosynthetic organisms can absorb 50% of incident solar radiation; photochemical cells could use materials absorbing a wider range of solar radiation. Plants have a theoretical threshold of 12% efficiency of glucose formation from photosynthesis, while a carbon reducing catalyst may have a lower threshold value. However, plants are efficient in using CO2 at atmospheric concentrations, something that artificial catalysts cannot perform yet.
The natural photosynthetic reaction consists of two half-reactions: oxidation and reduction. In plant photosynthesis, water molecules are photo-oxidized to release oxygen and protons. The second stage of plant photosynthesis is a light-independent reaction that converts carbon dioxide into glucose. This stage is also known as Calvin-Benson cycle:
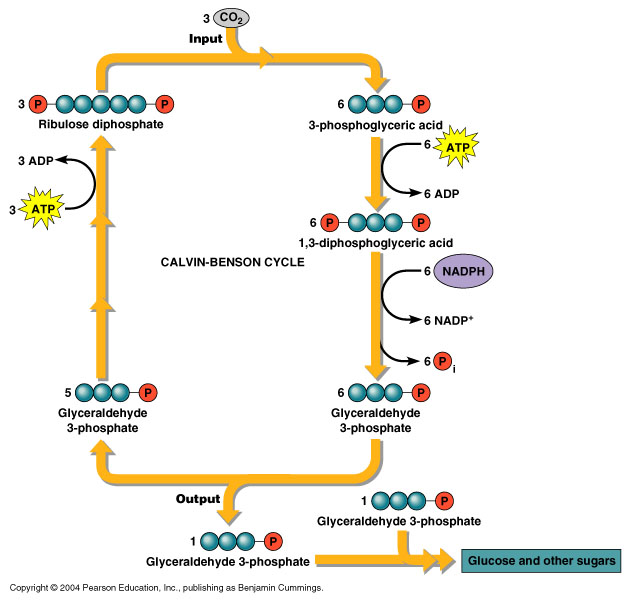
Recent studies are focused on photocatalysts to perform both of these reactions independently. Furthermore, the protons can be used for hydrogen production.
Photovoltaic energy is able to provide direct electrical current from sunlight but the main problem of photovoltaic energy is that the sunlight is neither constant nor homogeneous. Artificial photosynthesis aims to produce a fuel from sunlight that could be stored and used when sunlight was not available. In artificial photosynthesis only water and sunlight would be necessary for an environmental friendly energy production.The only by-product would be oxygen. Solar fuel seems to be cheapest than fossil fuels as gasoline.
One process for the creation of energy supply is the development of photocatalytic water splitting under solar radiation. The conversion of solar energy into hydrogen by means of a water-splitting process helped by photosemiconductor catalysts is one of the most promising technologies in development. This process has the potential to generate large quantities of hydrogen in an ecologically method. The conversion of solar energy into hydrogen (a clean fuel) under ambient conditions is one of the greatest challenges facing scientists at present.
Solar fuel cells for hydrogen production can be:
- A homogeneous system where catalysts are not separated: hydrogen and oxygen are produced in the same compartment. They compose an explosive mixture, demanding further gas purification. All components must be active in more or less the same conditions of pH, temperature, etc.
- A heterogeneous system has an anode and a cathode, making possible the separation of oxygen and hydrogen production. In this way, different components do not work necessarily in the same conditions. However, this system is more difficult to carry out and more expensive than the homogeneous system.
Photosynthetic microorganisms also known as microalgae and cyanobacteria can produce solar fuels. This field research is focusing its efforts to study the suitable selection and manipulation of these microorganisms. Several carbon-based biofuels have also been produced using cyanobacteria, such as 1-butanol.
Filamentous cyanobacteria are nitrogen-fixing microorganisms that possess the enzyme nitrogenase which is responsible for conversion of atmospheric N2 into ammmonia. Molecular hydrogen is a byproduct of this reaction, and is many times not released by the microorganism, but rather taken up by a hydrogen-oxidizing hydrogenase. One way of forcing these organisms to produce hydrogen is then to annihilate uptake hydrogenase activity. Synthetic biology techniques are predicted to be useful in this field.
Types of Processes
Artificial photosynthesis has the intention of constructing systems doing the same type of processes that the natural photosynthesis. One of the simplest designs is where the photosensitizer is linked in tandem between a water oxidation catalyst and a hydrogen catalyst:
- The photosensitizer transfers electrons to the hydrogen catalyst when light reaches it; the electrons becoming oxidized in the process.
- This drives the water splitting catalyst to donate electrons to the photosensitizer. The oxidized donor is able to perform water oxidation.

HYDROGEN CATALYSTS
Hydrogen is the simplest solar fuel to synthesize because it involves only the transference of two electrons to two protons.
The reaction is as follows:
2 e− + 2 H+ ↔ H+ + H− ↔ H2
The enzymes involved in the proton-to-hydrogen converting catalysts are hydrogenases. These can either reduce protons to molecular hydrogen or oxidize hydrogen to protons and electrons.
There is a lot of information related to the hydrogen catalyst and thanks to this information a big amount of molecules reproducing the structure of the active site of both nickel-iron and iron-iron hydrogenases have been synthesized.
Other catalysts are not structural mimics of hydrogenase but rather functional ones. Synthesized catalysts include structural H-cluster models,a dirhodium photocatalyst, and cobalt catalysts. There is another catalyst to produce solar fuel but it is more expensive and more difficult to carry out due to a higher temperature is required. This is the water-oxidizing catalysts.
Trends
Research in artificial photosynthesis is a multidisciplinary field. Some techniques employed in making and investigating catalysts and solar cells include organic and inorganic chemical synthesis, spectroscopic methods, electrochemistry, molecular biology, microbiology, synthetic biology and crystallography, among others.
Methodologies under development
PHOTOELECTROCHEMICALLS CELLS
Photoelectrochemical cells are heterogeneous systems that use light to produce electricity or hydrogen. Dye-sensitized solar cell is a hopeful, recent type of solar cell. This cell depends on a semiconductor for current conduction on one electrode and it is covered by a coating of an organic or inorganic dye that acts as a photosensitizer; the counter electrode is a platinum catalyst for H2 production. These cells have a self-repair mechanism and solar-to-electricity conversion efficiencies rivaling those of solid-state semiconductor ones.
PHOTOCATALYTIC WATER SPLITING IN HOMOGENEOUS SYSTEMS
Direct water oxidation by photocatalysts is a more efficient usage of solar energy than photoelectrochimical water splitting because it avoids an intermediate thermal or electrical energy conversion step. Some ruthenium complexes are able to oxidize water under solar light irradiation. They have photostability. Improvement of catalyst stability has been tried resorting to polyoxometalates, in particular ruthenium-based ones.
There are another emerging methodologies as the hydrogen-producing artificial systems and NADP+/NADPH coenzyme-inspired catalyst and photobiological production of fuels.
